Law of Inertia and Head Injuries
Mild Traumatic Brain Injury, Not So Mild After All
ScienceDaily (Feb. 22, 2010) — Douglas Smith, MD, director of the Center for Brain Injury and Repair and professor of Neurosurgery at the University of Pennsylvania School of Medicine, presented findings on the molecular mechanism at play in mild traumatic brain injury (mTBI), commonly known as concussions, recently at the 2010 American Association for the Advancement of Science meeting in San Diego.
Although mTBI affects over 1.7 million people each year in the United States, it is generally ignored as a major health issue. However, this “mild” form of injury induces persisting neurological and cognitive problems in many of these patients, exacting an enormous emotional and financial toll on society.
Despite the prevalence and impact of mTBI, little is known about how mTBI affects nerve cells and connections in the brain, and therefore clinical outcomes after injury. Smith and colleagues have begun to amass data from human and animal studies on mTBI at 2-4 days after injury using advanced neuroimaging techniques. They have found distinct changes throughout the white matter in the brain. Also, protein markers of brain pathology were identified after mTBI in the blood of mTBI patients.
Concussion, from the Latin concutere (“to shake violently”)[1] or concussus (“action of striking together”),[2] is the most common type of traumatic brain injury. The terms mild brain injury, mild traumatic brain injury (MTBI), mild head injury (MHI), minor head trauma, and concussion may be used interchangeably,[3][4] although the last is often treated as a narrower category.[5] Although the term “concussion” is still used in sports literature as interchangeable with “MHI” or “MTBI”, the general clinical medical literature now uses “MTBI” instead.[6] In this article, “concussion” and “MTBI” are used interchangeably. Frequently defined as a head injury
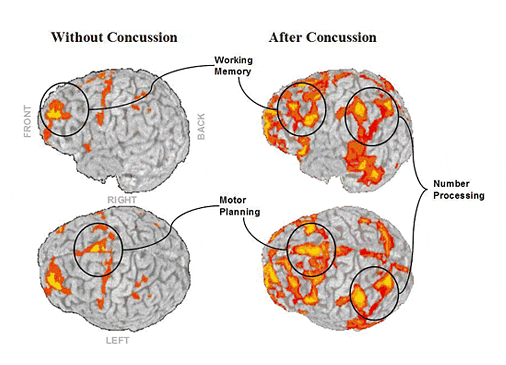
with a temporary loss of brain function, concussion causes a variety of physical, cognitive, and emotional symptoms, which may not be recognized if subtle.
Treatment involves monitoring as well as physical and cognitive rest (reduction of such activities as school work, playing video games and text messaging).[7] Symptoms usually resolve within three weeks, though they may persist or complications may occur for month or years.[8]
Those who have had one concussion seem more susceptible to another, especially if the new injury occurs before symptoms from the previous concussion have completely resolved.[9] There is also a negative progressive process in which smaller impacts cause the same symptom severity.[7] Repeated concussions may increase the risk in later life for dementia, Parkinson’s disease, and/or depression.[9]
A variety of signs accompany concussion including somatic (such as headache), cognitive (such as feeling in a fog), emotional (such as emotional changeability), physical signs (such as loss of consciousness or amnesia), behavioral changes (such as irritability), cognitive impairment (such as slowed reaction times), and/or sleep disturbances.[7] A 2010 Pediatrics review article focusing on children and adolescents noted that fewer than 10% of sports-related concussions had associated loss of consciousness.[10]
Due to varying definitions and possible underreporting, the rate at which concussion occurs annually is not accurately known, but is estimated to be more than 6 per 1,000 people.[11] Common causes include sports injuries, bicycle accidents, car accidents, and falls, the latter two being the most frequent among adults.[12] In addition to a blow to the head, concussion may be caused by acceleration forces without a direct impact, and on the battlefield, MTBI is a potential consequence of nearby explosions.[13]
It is not known whether the brain in concussion is structurally damaged or whether there is mainly a loss of function with only physiological changes.[14][needs update] Cellular damage has reportedly been found in concussed brains, but it may have been due to artifacts from the studies.[15] It is now thought that structural and neuropsychiatric factors may both be responsible for the effects of concussion.[16]
Rear Impact Collision and the Law of Inertia
Rear Impact Collisions are a major cause of concussion in the US, approximately 60% to 67% of all TBI are the result of MVC.
The Law Of Inertia
The object in question in a whiplash trauma is a human body. The human body has two large parts that have their own separate inertia, the trunk and the head. These two large pieces of inertial mass (the head and the trunk) are connected by a thin pole, the neck.
Upon impact, the target vehicle begins to move forward into the occupant, making contact chiefly through the seat back. In accordance with Newton’s 1st law of motion, the occupant’s inertia resists this motion.
As the seat back continues to move forward, the occupant must yield. Initially, the thoracic curve is flattened by the seat back. This results in a vertical compressive force which is transmitted through the spine.
So far, we have not been able to determine to what degree–if any–the lumbar spine also flattens. As the vertical compressive force (-z) continues up the spine, some rise of the torso also occurs. This is called ramping and is halted after 1-3 inches of vertical displacement, usually because of the restraining effect of the seat belt and the weight of the torso. Meanwhile, as the torso now is undergoing both a z acceleration vertical and an x linear acceleration, the head–also acting in accordance with Newton’s 1st law of motion–attempts to remain at rest. As the vertical force extends upwards into the neck it initiates flexion of the upper cervical segments and hyperextension of the lower segments.
As the torso continues to move forward, the neck begins to pull the head along with it. This has the effect of further flexing the upper cervical spine and hyperextending the lower cervical spine (primarily the C5-6 segments) and the spine assumes an s-shaped configuration, Figure below. The head also is induced to extend along with the neck as the head takes up the backset distance during the head lag phase.
Depending on specific head restraint geometry (occupant’s position relative to the restraint), head restraint contact will usually occur in about 100 msec at which time head translational acceleration will peak. Any stored energy in the seat back from its deflection (usually about 5-15 degrees) will be released as the occupant begins to more forward into the re-entry phase. This effectively increases the torso and head speed (overspeed).
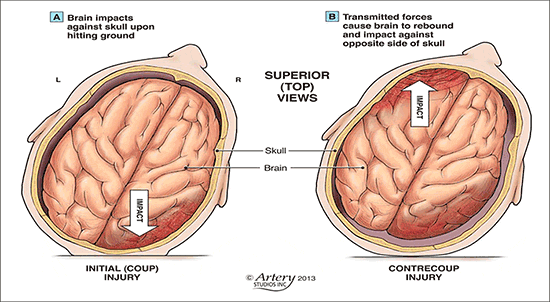
As this change of phase occurs, the direction of horizontal shear reverses rapidly and the rearward bending moment quickly gives way to a forward bending moment. Depending on the initial position of the occupant with respect to safety harnesses, the lap and shoulder portions will eventually restrain the forward moving torso which will effectively aggravate the neck’s bending moment since the forward moving head’s inertia again is behaving in accordance with Newton’s 1st law of motion–this time in the x direction with some angular momentum and acceleration. The weight of the human head (10-12 lbs.) plus the force of a moving vehicle that may carry 10,000 to 20,000 pounds of force is not a good combination. Below is the example of the head striking which is also the same mechanism of the head striking the head rest.
Post-traumatic amnesia is better predictor of outcome after MTBI than loss of consciousness; recent research has indicated that amnesia may be up to 4-10 times more predictive of symptoms and cognitive deficits following concussion than is LOC (less than 1 minute). CDC Toolkit, “Heads Up: Brain Injury in Your Practice,” Acute Concussion Evaluation (ACE), Instructions, p. 2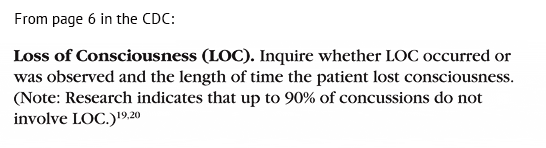
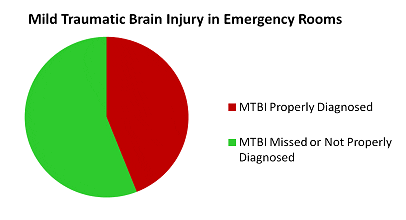
Emergency rooms miss MTBI 56% of time. Powell JM, Ferraro JV, Dikmen SS, Temkin NR, Bell KR, Accuracy of mild traumatic brain injury diagnosis. Arch Phys Med Rehabil. 2008 Aug;89(8):1550-5
This is a troubling trend, this also indicate that most general medical practices, family practices, primary care physicians, urgent care facilities and chiropractic offices are also missing at least 56% of the injuries.
Classification
No single definition of concussion, minor head injury,[17] or mild traumatic brain injury is universally accepted.[18] In 2001, the expert Concussion in Sport Group of the first International Symposium on Concussion in Sport[19] defined concussion as “a complex pathophysiological process affecting the brain, induced by traumatic biomechanical forces.”[20] It was agreed that concussion typically involves temporary impairment of neurological function that heals by itself within time, and that neuroimaging normally shows no gross structural changes to the brain as the result of the condition.[8]
However, although no structural brain damage occurs according to the classic definition,[21] some researchers have included injuries in which structural damage has occurred and the National Institute for Health and Clinical Excellence definition includes physiological or physical disruption in the brain’s synapses.[22] Also, by definition, concussion has historically involved a loss of consciousness. However, the definition has evolved over time to include a change in consciousness, such as amnesia,[23] although controversy continues about whether the definition should include only those injuries in which loss of consciousness occurs.[1] This debate resurfaces in some of the best-known concussion grading scales, in which those episodes involving loss of consciousness are graded as more being severe than those without.[24]
Definitions of mild traumatic brain injury (MTBI) were inconsistent until the World Health Organization’s International Statistical Classification of Diseases and Related Health Problems (ICD-10) provided a consistent, authoritative definition across specialties in 1992.[4] Since then, various organizations such as the American Congress of Rehabilitation Medicine[25] and the American Psychiatric Association in its Diagnostic and Statistical Manual of Mental Disorders[4] have defined MTBI using some combination of loss of consciousness (LOC), post-traumatic amnesia (PTA), and the Glasgow Coma Scale (GCS).
Concussion falls under the classification of mild TBI,[26] but it is not clear whether concussion is implied in mild brain injury or mild head injury.[27] “MTBI” and “concussion” are often treated as synonyms in medical literature[25] but other injuries such as intracranial hemorrhages (e.g. intra-axial hematoma, epidural hematoma, and subdural hematoma) are not necessarily precluded in MTBI[8] or mild head injury,[28][29] as they are in concussion.[30] MTBI associated with abnormal neuroimaging may be considered “complicated MTBI”.[31] “Concussion” can be considered to imply a state in which brain function is temporarily impaired and “MTBI” to imply a pathophysiological state, but in practice few researchers and clinicians distinguish between the terms.[8] Descriptions of the condition, including the severity and the area of the brain affected, are now used more often than “concussion” in clinical neurology.[32]
Concussion grading systems
At least 41 systems measure the severity, or grade, of a mild head injury,[8] and there is little agreement about which is best.[33] In an effort to simplify, the 2nd International Conference on Concussion in Sport, meeting in Prague in 2004, decided that these systems should be abandoned in favor of a ‘simple’ or ‘complex’ classification.[34] However, the 2008 meeting in Zurich abandoned the simple versus complex terminology, although the participants did agree to keep the concept that most (80–90%) concussions resolve in a short period (7–10 days), and although the recovery time frame may be longer in children and adolescents.[7]
In the past, the decision to allow athletes to return to participation was frequently based on the grade of concussion. However, current research and recommendations by professional organizations including the National Athletic Trainers’ Association recommend against such use of these grading systems. Currently, injured athletes are prohibited from returning to play before they are symptom-free during both rest and exertion and until results of the neuropsychological tests have returned to pre-injury levels.
Three grading systems have been most widely followed: by Robert Cantu, the Colorado Medical Society, and the American Academy of Neurology.[24] Each employs three grades:
Comparison of historic concussion grading scales – not currently recommended for use by medical professionals.
Grading Scales for Concussions: Grade I Grade II Grade III
Cantu
I. Post-traumatic amnesia <30 minutes, no loss of consciousness
II. Loss of consciousness <5 minutes or amnesia lasting 30 minutes–24 hours
III. Loss of consciousness >5 minutes or amnesia >24 hours
Colorado Medical Society
I. Confusion, no loss of consciousness
II. Confusion, post-traumatic amnesia, no loss of consciousness
III. Any loss of consciousness
American Academy of Neurology
I. Confusion, symptoms last <15 minutes, no loss of consciousness
II. Symptoms last >15 minutes, no loss of consciousness
III. Loss of consciousness (IIIa, coma lasts seconds, IIIb for minutes)
Signs and symptoms
Concussion is associated with a variety of symptoms, which typically occur rapidly after the injury.[19] Early symptoms usually subside within days or weeks.[15] The number and type of symptoms a person suffers varies widely.[18]
Physical
Headache is the most common MTBI symptom.[25] Others include dizziness, vomiting, nausea, lack of motor coordination, difficulty balancing,[25] or other problems with movement or sensation. Visual symptoms include light sensitivity,[35] seeing bright lights,[33] blurred vision,[15] and double vision.[36] Tinnitus, or a ringing in the ears, is also commonly reported.[15] In one in about seventy concussions, concussive convulsions occur, but seizures that take place during or immediately after concussion are not the same as post-traumatic seizures, and, unlike post-traumatic seizures, are not predictive of post-traumatic epilepsy, which requires some form of structural brain damage, not just a momentary disruption in normal brain functioning.[37] Concussive convulsions are thought to result from temporary loss or inhibition of motor function, and are not associated either with epilepsy or with more serious structural damage. They are not associated with any particular sequelae, and have the same high rate of favorable outcomes as concussions without convulsions.[38]
Cognitive and emotional
Cognitive symptoms include confusion, disorientation, and difficulty focusing attention. Loss of consciousness may occur, but is not necessarily correlated with the severity of the concussion if it is brief.[20] Post-traumatic amnesia, in which events following the injury cannot be recalled, is a hallmark of concussion.[25] Confusion, another concussion hallmark, may be present immediately or may develop over several minutes.[25] A person may repeat the same questions,[39] be slow to respond to questions or directions, have a vacant stare, or have slurred[25] or incoherent speech.[40] Other MTBI symptoms include changes in sleeping patterns[15] and difficulty with reasoning,[36] concentrating, and performing everyday activities.[25]
Affective results of concussion include crankiness, loss of interest in favorite activities or items,[41] tearfulness,[5] and displays of emotion that are inappropriate to the situation.[40] Common symptoms in concussed children include restlessness, lethargy, and irritability.[42]
Mechanism
Rotational force is key in concussion. Punches in boxing deliver more rotational force to the head than impacts in sports such as American football, and boxing carries a higher risk of concussion than football.[43]
The brain is surrounded by cerebrospinal fluid, which protects it from light[vague] trauma. More severe impacts, or the forces associated with rapid[vague] acceleration, may not be absorbed by this cushion.[14] Concussion may be caused by impact forces, in which the head strikes or is struck by something, or impulsive forces, in which the head moves without itself being subject to blunt trauma (for example, when the chest hits something and the head snaps forward).[5]
Forces may cause linear, rotational, or angular movement of the brain, or a combination of them.[5] In rotational movement, the head turns around its center of gravity, and in angular movement it turns on an axis not through its center of gravity.[5] The amount of rotational force is thought to be the major component in concussion[44] and its severity.[8] Studies with athletes have shown that the amount of force and the location of the impact are not necessarily correlated with the severity of the concussion or its symptoms, and have called into question the threshold for concussion previously thought to exist at around 70–75g.[45][46]
The parts of the brain most affected by rotational forces are the midbrain and diencephalon.[1][12] It is thought that the forces from the injury disrupt the normal cellular activities in the reticular activating system located in these areas, and that this disruption produces the loss of consciousness often seen in concussion.[12] Other areas of the brain that may be affected include the upper part of the brain stem, the fornix, the corpus callosum, the temporal lobe, and the frontal lobe.[47] Angular accelerations of 4600, 5900, or 7900 radian/s2 are estimated to have 25, 50, or 80% risk of MTBI respectively.[48]
Pathophysiology
In both animals and humans, MTBI can alter the brain’s physiology for hours to weeks, setting into motion a variety of pathological events.[49] As one example, in animal models, after an initial increase in glucose metabolism, there is a subsequent reduced metabolic state which may persist for up to four weeks after injury.[10] Though these events are thought to interfere with neuronal and brain function, the metabolic processes that follow concussion are reversible in a large majority of affected brain cells; however, a few cells may die after the injury.[31] Included in the cascade of events unleashed in the brain by concussion is impaired neurotransmission, loss of regulation of ions, deregulation of energy use and cellular metabolism, and a reduction in cerebral blood flow.[31] Excitatory neurotransmitters, chemicals such as glutamate that serve to stimulate nerve cells, are released in excessive amounts.[50] The resulting cellular excitation causes neurons to fire excessively.[51] This creates an imbalance of ions such as potassium and calcium across the cell membranes of neurons (a process like excitotoxicity).[31] At the same time, cerebral blood flow is relatively reduced for unknown reasons,[35] though the reduction in blood flow is not as severe as it is in ischemia.[31] Thus cells get less glucose than they normally do, which causes an “energy crisis”.[35]”
Concurrently with these processes, the activity of mitochondria may be reduced, which causes cells to rely on anaerobic metabolism to produce energy, increasing levels of the byproduct lactate.[31]
For a period of minutes to days after a concussion, the brain is especially vulnerable to changes in intracranial pressure, blood flow, and anoxia.[35] According to studies performed on animals (which are not always applicable to humans), large numbers of neurons can die during this period in response to slight, normally innocuous changes in blood flow.[35]
Concussion involves diffuse (as opposed to focal) brain injury, meaning that the dysfunction occurs over a widespread area of the brain rather than in a particular spot.[52] Concussion is thought to be a milder type of diffuse axonal injury, because axons may be injured to a minor extent due to stretching.[5] Animal studies in which primates were concussed have revealed damage to brain tissues such as small petechial hemorrhages and axonal injury.[53] Axonal damage has been found in the brains of concussion sufferers who died from other causes, but inadequate blood flow to the brain due to other injuries may have contributed to the damage.[15] Findings from a study of the brains of dead NFL athletes who received concussions suggest that lasting damage is done by such injuries. This damage, the severity of which increases with the cumulative number of concussions sustained, can lead to a variety of other health issues.[54]
The debate over whether concussion is a functional or structural phenomenon is ongoing.[55] Structural damage has been found in the mildly traumatically injured brains of animals, but it is not clear whether these findings would apply to humans.[1] Such changes in brain structure could be responsible for certain symptoms such as visual disturbances, but other sets of symptoms, especially those of a psychological nature, are more likely to be caused by reversible pathophysiological changes in cellular function that occur after concussion, such as alterations in neurons’ biochemistry.[8] These reversible changes could also explain why dysfunction is frequently temporary.[55] A task force of head injury experts called the Concussion In Sport Group met in 2001 and decided that “concussion may result in neuropathological changes but the acute clinical symptoms largely reflect a functional disturbance rather than structural injury.”[19]
In summary, and extrapolating from animal studies, the pathology of a concussion seems to start with the disruption of the cell membrane of nerve cells. This results in a migration of potassium from within the cell into the extracellular space with subsequent release of glutamate which potentiates further potassium shift, in turn resulting in depolarization and suppression of nerve activity. In an effort to restore ion balance, the sodium-potassium ion pumps increase activity, which results in excessive ATP (adenosine triphosphate) consumption and glucose utilization. Lactate accumulates but, paradoxically, cerebral blood flow decreases, which leads to a proposed “energy crisis.” After this increase in glucose metabolism, there is a subsequent lower metabolic state which may persist for up to 4 weeks after injury. A completely separate pathway involves a large amount of calcium accumulating in cells, which may impair oxidative metabolism and begin further biochemical pathways that result in cell death. Again, both of these main pathways have been established from animal studies and the extent to which they apply to humans it is still somewhat unclear.[10]
Diagnosis
Health care providers examine head trauma recipients to ensure that the injury is not a more severe emergency such as an intracranial hemorrhage. Assessment includes the “ABCs” (airway, breathing, circulation) and stabilization of the cervical spine which is assumed to be injured in any athlete who is found to be unconscious after head or neck injury. Indications that screening for more serious injury is needed include worsening of symptoms such as headache, persistent vomiting,[56] increasing disorientation or a deteriorating level of consciousness,[57] seizures, and unequal pupil size.[58] Those with such symptoms, or those who are at higher risk for a more serious brain injury, may undergo brain imaging to detect lesions and are frequently observed for 24 – 48 hours.
Unequal pupil size is a sign of a brain injury more serious than concussion.

Diagnosis of MTBI is based on physical and neurological examination findings, duration of unconsciousness (usually less than 30 minutes) and post-traumatic amnesia (PTA; usually less than 24 hours), and the Glasgow Coma Scale (MTBI sufferers have scores of 13 to 15).[59] Neuropsychological tests exist to measure cognitive function and the international consensus meeting in Zurich recommended the use of the SCAT2 test.[15] [7] Such tests may be administered hours, days, or weeks after the injury, or at different times to demonstrate any trend.[60] Increasingly, athletes are also being tested pre-season to provide a baseline for comparison in the event of an injury, though this may not reduce risk or affect return to play.[61][62]
If the Glasgow Coma Scale is less than 15 at two hours, or less than 14 at any time, a CT is recommended.[12] In addition, a CT scan is more likely to be performed if observation after discharge is not assured or intoxication is present, there is suspected increased risk for bleeding, age greater than 60,[12] or less than 16. Most concussions, without complication, cannot be detected with MRI or CT scans.[44] However, changes have been reported to show up on MRI and SPECT imaging in concussed people with normal CT scans, and post-concussion syndrome may be associated with abnormalities visible on SPECT and PET scans.[31] Mild head injury may or may not produce abnormal EEG readings.[63]
Concussion may be under-diagnosed. The lack of the highly noticeable signs and symptoms may lead clinicians to miss the injury, and athletes may minimize their injuries to remain in the competition.[6] A retrospective survey in 2005 suggested that more than 88% of concussions are unrecognized.[64]
Diagnosis of concussion can be complicated because it shares symptoms with other conditions. For example, post-concussion symptoms such as cognitive problems may be misattributed to brain injury when they are in fact due to post-traumatic stress disorder (PTSD).[65]
As a clinician you are responsible for your patient and with over 1.7 million recorded concussions in the US every year and more than 56% are missed by the medical community, we need to be prudent and consistent with our testing.
[wc_fa icon=”ellipsis-h” margin_left=”” margin_right=””][/wc_fa] The Personal Injury Institute – Personal Injury Report is a monthly publication by me, Dr. Matthew J. DeGaetano, DC, Certified in Whiplash and Brain Traumatology and Colossus. I am a 1997 graduate of Parker College of Chiropractic. I have managed about 6,000 whiplash injury cases in NY and Texas in the past 17 years. I am the personal consultant for over 300 offices and over 100 personal injury law firms nationally.
The purpose of The Personal Injury Institute – Personal Injury Report is to keep you updated on relevant academic concepts pertaining to whiplash injury patients. I hope that the information is useful in terms of enhanced understanding, as well as helpful for the personal injury attorneys to deal with insurance claim adjusters, dealing with Colossus systems and adverse medical experts.
Our clinics are well informed and trained in these concepts of personal injury and the details of Colossus and will be a valuable asset in personal injury cases, in terms of both academics and treatment. Additionally, expert chiropractors and your law firm will have access to daily phone consultation with me, to discuss any pertinent issues faced by them, on a particular case.
I hope that you find this Report as a valuable resource.
Sincerely,
Matthew J. DeGaetano, DC
References:
1. Pearce JM (2007). “Observations on concussion. A review”. European Neurology 59 (3–4): 113–9. doi:10.1159/000111872. PMID 18057896.
2. Brooks D, Hunt B (2006). “Current concepts in concussion diagnosis and management in sports: A clinical review”. BC Medical Journal 48 (9): 453–459.
3. National Center for Injury Prevention and Control (2003). “Report to congress on mild traumatic brain injury in the United States: Steps to prevent a serious public health problem” (PDF). Atlanta, GA: Centers for Disease Control and Prevention. Retrieved 2008-01-19.
4. Petchprapai N, Winkelman C (2007). “Mild traumatic brain injury: determinants and subsequent quality of life. A review of the literature”. Journal of Neuroscience Nursing 39 (5): 260–72. PMID 17966292.
5. Sivák Š, Kurča E, Jančovič D, Petriščák Š, Kučera P (2005). “Nácrt súcasného pohl’adu na problematiku l’ahkých poranení mozgu so zameraním na dospelú populáciu” [An outline of the current concepts of mild brain injury with emphasis on the adult population] (PDF). Časopis Lėkařů Českých (in Slovak) 144 (7): 445–450. PMID 16161536.
6. Barth JT, Varney NR, Ruchinskas RA, Francis JP (1999). “Mild head injury: The new frontier in sports medicine”. In Varney NR, Roberts RJ. The Evaluation and Treatment of Mild Traumatic Brain Injury. Hillsdale, New Jersey: Lawrence Erlbaum Associates. pp. 85–6. ISBN 0-8058-2394-8. Retrieved 2008-03-06.
7. McCrory, P; Meeuwisse, W; Johnston, K; Dvorak, J; Aubry, M; Molloy, M; Cantu, R (2009 Jul-Aug). “Consensus statement on concussion in sport: the 3rd International Conference on Concussion in Sport held in Zurich, November 2008.”. Journal of athletic training 44 (4): 434–48. doi:10.4085/1062-6050-44.4.434. PMC 2707064. PMID 19593427.
8. Anderson T, Heitger M, Macleod AD (2006). “Concussion and mild head injury”. Practical Neurology 6 (6): 342–57. doi:10.1136/jnnp.2006.106583.
9. Kenneth Maiese (January 2008). “Concussion”. The Merck Manual Home Health Handbook.
10. From American Academy of Pediatrics, Clinical Report, “Sport-Related Concussion in Children and Adolescents”, Pediatrics, Mark E. Halstead, MD, Kevin D. Walter, MD, The Council on Sports Medicine and Fitness, Vol. 126 No. 3, September 1, 2010.
11. Cassidy JD, Carroll LJ, Peloso PM, Borg J, von Holst H, Holm L, et al. (2004). “Incidence, risk factors and prevention of mild traumatic brain injury: Results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury”. Journal of Rehabilitation Medicine 36 (Supplement 43): 28–60. doi:10.1080/16501960410023732. PMID 15083870.
12. Ropper AH, Gorson KC (2007). “Clinical practice. Concussion”. New England Journal of Medicine 356 (2): 166–72. doi:10.1056/NEJMcp064645. PMID 17215534.
13. Warden, Deborah L.; French, Louis M.; Shupenko, Leslie; Fargus, Jamie; Riedy, Gerard; Erickson, Marleigh E.; Jaffee, Michael S.; Moore, David F. (2009). “Case report of a soldier with primary blast brain injury”. NeuroImage 47: T152–3. doi:10.1016/j.neuroimage.2009.01.060. PMID 19457364.
14. Shaw NA (2002). “The neurophysiology of concussion”. Progress in Neurobiology 67 (4): 281–344. doi:10.1016/S0301-0082(02)00018-7. PMID 12207973.
15. Rees PM (2003). “Contemporary issues in mild traumatic brain injury”. Archives of Physical Medicine and Rehabilitation 84 (12): 1885–94. doi:10.1016/j.apmr.2003.03.001. PMID 14669199.
16. Riggio S, Wong M (2009 Apr). “Neurobehavioral sequelae of traumatic brain injury”. Mt Sinai J Med 76 (2): 163–72. doi:10.1002/msj.20097. PMID 19306386.
17. Satz P, Zaucha K, McCleary C, Light R, Asarnow R, Becker D (1997). “Mild head injury in children and adolescents: A review of studies (1970–1995)”. Psychological Bulletin 122 (2): 107–131. doi:10.1037/0033-2909.122.2.107. PMID 9283296.
18. Comper P, Bisschop SM, Carnide N, Tricco A (2005). “A systematic review of treatments for mild traumatic brain injury”. Brain Injury 19 (11): 863–880. doi:10.1080/02699050400025042. ISSN 0269-9052. PMID 16296570.
19. Aubry M, Cantu R, Dvorak J, Graf-Baumann T, Johnston K, Kelly J, Lovell M, McCrory P, Meeuwisse W, Schamasch P; Concussion in Sport Group. (2002). “Summary and agreement statement of the first International Conference on Concussion in Sport, Vienna 2001*”. British Journal of Sports Medicine 36 (1): 6–10. PMC 1724447. PMID 11867482.
20. Cantu RC (2006). “An overview of concussion consensus statements since 2000” (PDF). Neurosurgical Focus 21 (4:E3): 1–6.
21. Parkinson D (1999). “Concussion confusion”. Critical Reviews in Neurosurgery 9 (6): 335–339. doi:10.1007/s003290050153. ISSN 1433-0377.
22. Head Injury: Triage, Assessment, Investigation and Early Management of Head Injury in Infants, Children and Adults (PDF). National Institute for Health and Clinical Excellence. September 2007. ISBN 0-9549760-5-3. Retrieved 2008-01-26.
23. Ruff RM, Grant I (1999). “Postconcussional disorder: Background to DSM-IV and future considerations”. In Varney NR, Roberts RJ. The Evaluation and Treatment of Mild Traumatic Brain Injury. Hillsdale, New Jersey: Lawrence Erlbaum Associates. p. 320. ISBN 0-8058-2394-8.
24. Cobb S, Battin B (2004). “Second-impact syndrome”. The Journal of School Nursing 20 (5): 262–7. doi:10.1177/10598405040200050401. PMID 15469376.
25. Kushner D (1998). “Mild Traumatic brain injury: Toward understanding manifestations and treatment”. Archives of Internal Medicine 158 (15): 1617–1624. doi:10.1001/archinte.158.15.1617. PMID 9701095.
26. Lee LK (2007). “Controversies in the sequelae of pediatric mild traumatic brain injury”. Pediatric Emergency Care 23 (8): 580–583. doi:10.1097/PEC.0b013e31813444ea. PMID 17726422.
27. Benton AL, Levin HS, Eisenberg HM (1989). Mild Head Injury. Oxford [Oxfordshire]: Oxford University Press. pp. v. ISBN 0-19-505301-X.
28. van der Naalt J (2001). “Prediction of outcome in mild to moderate head injury: A review”. Journal of Clinical and Experimental Neuropsychology 23 (6): 837–851. doi:10.1076/jcen.23.6.837.1018. PMID 11910548
29. Savitsky EA, Votey SR (2000). “Current controversies in the management of minor pediatric head injuries”. American Journal of Emergency Medicine 18 (1): 96–101. doi:10.1016/S0735-6757(00)90060-3. PMID 10674544.
30. Parikh S, Koch M, Narayan RK (2007). “Traumatic brain injury”. International Anesthesiology Clinics 45 (3): 119–135. doi:10.1097/AIA.0b013e318078cfe7. PMID 17622833.
31. Iverson GL (2005). “Outcome from mild traumatic brain injury”. Current Opinion in Psychiatry 18 (3): 301–17. doi:10.1097/01.yco.0000165601.29047.ae. PMID 16639155.
32. Larner AJ, Barker RJ, Scolding N, Rowe D (2005). The A-Z of Neurological Practice: a Guide to Clinical Neurology. Cambridge, UK: Cambridge University Press. p. 199. ISBN 0-521-62960-8.
33. Cantu RC (2001). “Posttraumatic Retrograde and Anterograde Amnesia: Pathophysiology and Implications in Grading and Safe Return to Play”. Journal of Athletic Training 36 (3): 244–8. PMC 155413. PMID 12937491.
34. McCrory P, Johnston K, Meeuwisse W, Aubry M, Cantu R, Dvorak J, Graf-Baumann T, Kelly J, Lovell M, Schamasch P (2005 Apr). “Summary and agreement statement of the 2nd International Conference on Concussion in Sport, Prague 2004”. Br J Sports Med. 39(4) (4): 196–204. doi:10.1136/bjsm.2005.018614. PMC 1725173. PMID 15793085.
35. Bowen AP (2003). “Second impact syndrome: A rare, catastrophic, preventable complication of concussion in young athletes”. Journal of Emergency Nursing 29 (3): 287–9. doi:10.1067/men.2003.90. PMID 12776088.
36. Erlanger DM, Kutner KC, Barth JT, Barnes R (1999). “Neuropsychology of sports-related head enjury: Dementia pugilistica to post concussion syndrome”. The Clinical Neuropsychologist 13 (2): 193–209. doi:10.1076/clin.13.2.193.1963. PMID 10949160.
37. McCrory PR, Berkovic SF (1998). “Concussive convulsions. Incidence in sport and treatment recommendations”. Sports Medicine 25 (2): 131–6. PMID 9519401.
38. Perron AD, Brady WJ, Huff JS (2001). “Concussive convulsions: Emergency department assessment and management of a frequently misunderstood entity”. Academic Emergency Medicine 8 (3): 296–8. doi:10.1111/j.1553-2712.2001.tb01312.x. PMID 11229957.
39. Quality Standards Subcommittee of the American Academy of Neurology (1997). “Practice Parameter: The Management of Concussion in Sports (Summary Statement)” (PDF). American Academy of Neurology. pp. 1–7. Retrieved 2008-03-05.
40. Anderson MK, Hall SJ, Martin M (2004). Foundations of Athletic Training: Prevention, Assessment, and Management. Lippincott Williams & Wilkins. p. 236. ISBN 0-7817-5001-6. Retrieved 2008-01-09.
41. Mayo Clinic Staff (2007). “Concussio”. Mayo Clinic. Retrieved 2008-01-10.
42. Heegaard W, Biros M (2007). “Traumatic brain injury”. Emergency Medicine Clinics of North America 25 (3): 655–678, viii. doi:10.1016/j.emc.2007.07.001. PMID 17826211.
43. Pellman EJ, Viano DC (2006). “Concussion in professional football: Summary of the research conducted by the National Football League’s Committee on Mild Traumatic Brain Injury” (PDF). Neurosurgical Focus 21 (4): E12. PMID 17112190.
44. Poirier MP (2003). “Concussions: Assessment, management, and recommendations for return to activity”. Clinical Pediatric Emergency Medicine 4 (3): 179–85. doi:10.1016/S1522-8401(03)00061-2.
45. Guskiewicz KM, Mihalik JP, Shankar V, et al. (2007). “Measurement of head impacts in collegiate football players: Relationship between head impact biomechanics and acute clinical outcome after concussion”. Neurosurgery 61 (6): 1244–52; discussion 1252–3. doi:10.1227/01.neu.0000306103.68635.1a. PMID 18162904.
46. Gever D (December 7, 2007). “Any football helmet hit can cause potential concussion”. MedPage Today. Retrieved 2008-02-27.
47. Bigler ED (2008). “Neuropsychology and clinical neuroscience of persistent post-concussive syndrome”. Journal of the International Neuropsychological Society 14 (1): 1–22. doi:10.1017/S135561770808017X. PMID 18078527.
48. P Rousseau, A Post, T B Hoshizaki (2009). “The effects of impact management materials in ice hockey helmets on head injury criteria”. Proceedings of the Institution of Mechanical Engineers, Part P: Journal of Sports Engineering and Technology 223 (4): 159–65. doi:10.1243/17543371JSET36. ISSN 1754-3371.
49. McAllister TW, Sparling MB, Flashman LA, Saykin AJ (2001). “Neuroimaging findings in mild traumatic brain injury”. Journal of Clinical and Experimental Neuropsychology 23 (6): 775–91. doi:10.1076/jcen.23.6.775.1026. PMID 11910544.
50. Herring SA, Bergfeld JA, Boland A, Boyajian-O’Neil LA, Cantu RC, Hershman E, et al. (2005). “Concussion (mild traumatic brain injury) and the team physician: A consensus statement” (PDF). Medicine and Science in Sports and Exercise (American College of Sports Medicine, American Academy of Family Physicians, American Academy of Orthopaedic Surgeons, American Medical Society for Sports Medicine, American Orthopaedic Society for Sports Medicine, American Osteopathic Academy of Sports Medicine) 37 (11): 2012–6. doi:10.1249/01.mss.0000186726.18341.70. PMID 16286874.
51. Giza CC, Hovda DA (2001). “The Neurometabolic Cascade of Concussion”. Journal of Athletic Training 36 (3): 228–35. PMC 155411. PMID 12937489.
52. Hardman JM, Manoukian A (2002). “Pathology of head trauma”. Neuroimaging Clinics of North America 12 (2): 175–87, vii. doi:10.1016/S1052-5149(02)00009-6. PMID 12391630.
53. Hall RC, Hall RC, Chapman MJ (2005). “Definition, diagnosis, and forensic implications of postconcussional syndrome”. Psychosomatics 46 (3): 195–202. doi:10.1176/appi.psy.46.3.195. PMID 15883140.
54. CNN (2009-01-27). “Dead Athletes Brains Show Damage From Concussions”. Boston University: Center for the Study of Traumatic Encephalopathy. Retrieved 2009-01-28.
55. McCrory PR, Berkovic SF (2001). “Concussion: The history of clinical and pathophysiological concepts and misconceptions”. Neurology 57 (12): 2283–89. doi:10.1212/WNL.57.12.2283. PMID 11756611.
56. Cook RS, Schweer L, Shebesta KF, Hartjes K, Falcone RA (2006). “Mild traumatic brain injury in children: Just another bump on the head?”. Journal of Trauma Nursing 13 (2): 58–65. PMID 16884134.
57. Kay A, Teasdale G (2001). “Head injury in the United Kingdom”. World Journal of Surgery 25 (9): 1210–20. doi:10.1007/s00268-001-0084-6. PMID 11571960.
58. “Facts About Concussion and Brain Injury”. Centers for Disease Control and Prevention. 2006. Retrieved 2008-01-13.
59. Borg J, Holm L, Cassidy JD, et al. (2004). “Diagnostic procedures in mild traumatic brain injury: Results of the WHO collaborating centre task force on mild traumatic brain injury”. Journal of Rehabilitation Medicine 36 (Supplement 43): 61–75. doi:10.1080/16501960410023822. PMID 15083871.
60. Moser RS, Iverson GL, Echemendia RJ, Lovell MR, Schatz P, Webbe FM et al. (2007). “Neuropsychological evaluation in the diagnosis and management of sports-related concussion”. Archives of Clinical Neuropsychology 22 (8): 909–16. doi:10.1016/j.acn.2007.09.004. PMID 17988831.
61. Maroon JC, Lovell MR, Norwig J, Podell K, Powell JW, Hartl R (2000). “Cerebral concussion in athletes: Evaluation and neuropsychological testing”. Neurosurgery 47 (3): 659–69; discussion 669–72. doi:10.1097/00006123-200009000-00027. PMID 10981754.
62. Randolph C (2011). “Baseline neuropsychological testing in managing sport-related concussion: does it modify risk?”. Curr Sports Med Rep 10 (1): 21–26. doi:10.1249/JSR.0b013e318207831d. PMID 21228656.
63. Binder LM (1986). “Persisting symptoms after mild head injury: A review of the postconcussive syndrome”. Journal of Clinical and Experimental Neuropsychology 8 (4): 323–46. doi:10.1080/01688638608401325. PMID 3091631.
64. Delaney JS, Abuzeyad F, Correa JA, Foxford R (2005). “Recognition and characteristics of concussions in the emergency department population”. Journal of Emergency Medicine 29 (2): 189–97. doi:10.1016/j.jemermed.2005.01.020. PMID 16029831.
65. Bryant RA (2008). “Disentangling mild traumatic brain injury and stress reactions”. New England Journal of Medicine 358 (5): 525–7. doi:10.1056/NEJMe078235. PMID 18234757.
Jump up ^ Levy ML, Ozgur BM, Berry C, Aryan HE, Apuzzo ML (2004). “Birth and evolution of the football helmet”. Neurosurgery 55 (3): 656–61; discussion 661–2. doi:10.1227/01.NEU.0000134599.01917.AA. PMID 15335433.
66. Willer B, Leddy JJ (2006). “Management of concussion and post-concussion syndrome”. Current Treatment Options in Neurology 8 (5): 415–26. doi:10.1007/s11940-006-0031-9. ISSN 1534-3138. PMID 16901381.
67. Reymond MA, Marbet G, Radü EW, Gratzl O (1992). “Aspirin as a risk factor for hemorrhage in patients with head injuries”. Neurosurgical Review 15 (1): 21–5. doi:10.1007/BF02352062. PMID 1584433.
68. “What Can I Do to Help Feel Better After a Concussion?”. Atlanta, GA: Center of Disease Control. March 8, 2010.
Jump up ^ Thornton K, Carmody D (2008). “Efficacy of traumatic brain injury rehabilitation: interventions of QEEG-guided biofeedback, computers, strategies, and medications”. Applied Psychophysiology and Biofeedback 33 (2): 101–124. doi:10.1007/s10484-008-9056-z.
69. Anderson MK (2003). Fundamentals of sports injury management. Lippincott Williams & Wilkins. p. 79. ISBN 0-7817-3272-7.
70. “Information about NICE clinical guideline” (PDF). National Institute for Health and Clinical Excellence. September 2007. Retrieved 2008-01-26.
71. Murray ED, Buttner N, Price BH (2012). “Depression and Psychosis in Neurological Practice”. In Bradley WG, Daroff RB, Fenichel GM, Jankovic J. Bradley’s neurology in clinical practice. 1 (6th ed.). Philadelphia, PA: Elsevier/Saunders. p. 111. ISBN 1-4377-0434-4.
72. Alexander MP (1995). “Mild traumatic brain injury: Pathophysiology, natural history, and clinical management”. Neurology 45 (7): 1253–60. doi:10.1212/WNL.45.7.1253. PMID 7617178.
73. Masferrer R, Masferrer M, Prendergast V, Harrington TR (2000). “Grading scale for cerebral concussions”. BNI Quarterly (Barrow Neurological Institute) 16 (1). ISSN 0894-5799.
74. Schulz MR, Marshall SW, Mueller FO, et al. (November 2004). “Incidence and risk factors for concussion in high school athletes, North Carolina, 1996–1999”. American Journal of Epidemiology 160 (10): 937–44. doi:10.1093/aje/kwh304. PMID 15522850.
75. De Beaumont, Louis; Theoret, Hugo; Mongeon, David; Messier, Julie; Leclerc, Suzanne; Tremblay, Sebastien; Ellemberg, Dave; Lassonde, Maryse (2009). “Brain function decline in healthy retired athletes who sustained their last sports concussion in early adulthood”. Brain 132 (Pt 3): 695–708. doi:10.1093/brain/awn347. PMID 19176544.
76. Ryan LM, Warden DL (2003). “Post concussion syndrome”. International Review of Psychiatry 15 (4): 310–316. doi:10.1080/09540260310001606692. PMID 15276952.
77. Boake C, McCauley SR, Levin HS, Pedroza C, Contant CF, Song JX, et al. (2005). “Diagnostic criteria for postconcussional syndrome after mild to moderate traumatic brain injury”. Journal of Neuropsychiatry and Clinical Neurosciences 17 (3): 350–6. doi:10.1176/appi.neuropsych.17.3.350. PMID 16179657.
78. Harmon KG (1999). “Assessment and management of concussion in sports”. American Family Physician 60 (3): 887–892, 894. PMID 10498114.
79. Cantu RC (2007). “Chronic traumatic encephalopathy in the National Football League”. Neurosurgery 61 (2): 223–5. doi:10.1227/01.NEU.0000255514.73967.90. PMID 17762733.
80. Mendez MF (1995). “The neuropsychiatric aspects of boxing”. International Journal of Psychiatry in Medicine 25 (3): 249–62. doi:10.2190/CUMK-THT1-X98M-WB4C. PMID 8567192
81. Jordan BD (2000). “Chronic traumatic brain injury associated with boxing”. Seminars in Neurology 20 (2): 179–85. doi:10.1055/s-2000-9826. PMID 10946737.


